
Of course, we looked at if, with longer follow-up, we found any additional safety signals. That is PFS on second-line therapy or death, in patients randomized on the study. What we looked at was OS, PFS, and a new endpointa planned exploratory endpoint of PFS2. Now we have a median follow-up of almost 2 years.
So, the initial follow-up was under a year when the initial results were published and presented.
#KEYNOTE 189 UPDATE#
Gadgeel: At this year's ASCO, we provided an update with a longer follow-up. TARGETED ONCOLOGY: Can you explain the study updates that were presented at ASCO? And what the initial results showed was that there was a significant improvement in overall survival as well as progression-free survival with the addition of pembrolizumab to chemotherapy. The initial results of the study were first presented at AACR 2018 and simultaneously published in the New England Journal of Medicine. In this trial, over 600 patients were randomized to either pembrolizumab with chemotherapy or chemotherapy alone. Gadgeel: KEYNOTE-189 was a large, randomized phase III study evaluating the addition of pembrolizumab to chemotherapy of pemetrexed and a platinum in patients with metastatic nonsquamous NSCLC. TARGETED ONCOLOGY: Can you provide background on the KEYNOTE-189 study?
#KEYNOTE 189 TRIAL#
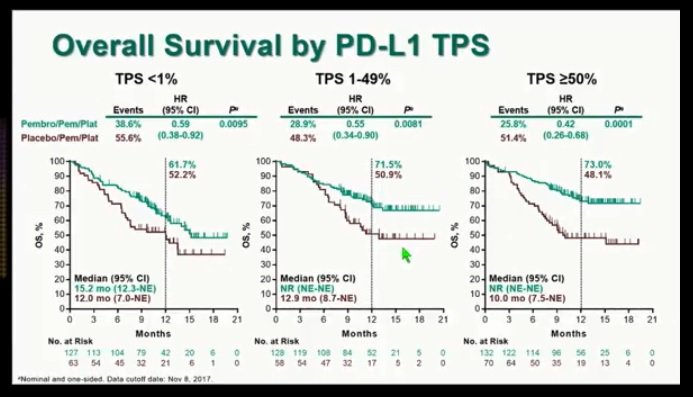
In an interview with Targeted Oncology,Shirish Gadgeel, MBBS, of the Department of Medicine, Hematology/Oncology Division, University of Michigan, discussed recent study updates from the phase III KEYNOTE-189 trial and potential implications for the combination on clinical practice. Additionally, the investigators noted that safety and tolerability remained manageable with the combination even with longer follow-up. The researchers confirmed that pembrolizumab can maximize outcomes when administered as part of first-line therapy in patients with metastatic nonsquamous NSCLC, with or without PD-L1 expression. The group of patients with negative PD-L1 expression (TPS <1% n = 190) showed an HR for OS of 0.52, HR for PFS of 0.63, and an HR for PFS2 of 0.46 the ORR was 32.3% versus 14.3% for patients who received the combination and chemotherapy alone, respectively. The group of patients with a TPS between 1% and 49% (n = 186) showed an HR for OS of 0.62, an HR for PFS of 0.51, and an HR for PFS2 of 0.59 the ORR in this subgroup was 49.2% with added pembrolizumab compared with 20.7% without.
#KEYNOTE 189 PLUS#
Pembrolizumab plus chemotherapy overall provided longer OS compared with chemotherapy alone in the overall population with a median OS of 22.0 months versus 10.7 months, respectively (hazard ratio, 0.56 95% CI, 0.45-0.70, P50% n = 202), the results showed a an HR for OS of 0.59, HR for PFS of 0.36, and an HR for PFS2 of 0.47 additionally, the objective response rate (ORR) was 62.1% for patients with high PD-L1 expression who received pembrolizumab plus chemotherapy compared with 24.3% in patients who received chemotherapy alone. About 45% of the patients in the pembrolizumab plus chemotherapy arm received a second-line therapy while about 60% of the patients in the placebo plus chemotherapy arm received a subsequent therapy, including immunotherapy. PFS2 was determined from the time of randomization with first-line therapy until patients progressed on their next-line treatment.


The researchers gathered outcomes data and information on post-study anticancer therapy.
In the occurrence of progressive disease, study subjects receiving chemotherapy were allowed to receive pembrolizumab. Patients were given 4 cycles of treatment followed by pembrolizumab or placebo was with maintenance pemetrexed. Patients were randomized 2:1 in the phase III trial, receiving either pembrolizumab (n = 410) or placebo (n = 206) plus pemetrexed and carboplatin or cisplatin. During the 2019 ASCO Annual Meeting, updates to the study were presented, showing a longer follow-up and progression-free survival 2 (PFS2) findings. The phase III KEYNOTE-189 trial led to the FDA approval of the combination of pembrolizumab (Keytruda) plus chemotherapy for the treatment of patients with metastatic nonsquamous nonsmall cell lung cancer (NSCLC) without EGFRor ALKalterations.

